How much time passes between your operators recording a weight and that weight entering your system?
If the answer is anything more than a few seconds, your facility has a contemporaneous documentation cannabis GMP gap that inspectors are trained to find. The Contemporaneous principle is the third pillar of the ALCOA++ data integrity framework, and it requires that data be recorded at the time the activity is performed, not minutes or hours afterward. Among all ten ALCOA++ principles, this is the one where the gap between paper-based (or desktop-only) workflows and mobile digital capture is most dramatic.
This guide defines what contemporaneous means in the regulatory context, explains why it matters specifically for cannabis manufacturing under Health Canada’s Good Production Practices (GPP), identifies the common workflow failures that violate this principle, and outlines how mobile data capture on the production floor addresses the problem at its root.
What Does Contemporaneous Mean in ALCOA++ Data Integrity?
The answer is straightforward: data must be recorded when the activity happens.
The Pharmaceutical Inspection Co-operation Scheme (PIC/S), whose member authorities include Health Canada, defines the Contemporaneous principle in PI 041-1: Good Practices for Data Management and Integrity as follows: “Data should be recorded at the time the activity is performed.” The guidance further states that backdating or post-hoc recording undermines the integrity of the record.
This definition is deceptively simple. It does not say data should be recorded “reasonably soon” or “by end of shift.” It says at the time of the activity. The implication is that any workflow introducing a delay between performing an action (weighing a batch, conducting an inspection, transferring inventory) and recording that action in the system of record creates a contemporaneous gap. The longer the delay, the greater the risk that the recorded data diverges from what actually happened.
The WHO TRS 996 Annex 5 reinforces this position by requiring that “data should be recorded contemporaneously, i.e., at the time the activity is performed.” The principle applies equally to paper and electronic records: whatever medium you use, the timestamp should reflect reality.
Why Does Contemporaneous Documentation Matter for Cannabis GMP?
Because Health Canada inspectors examine the relationship between when activities occurred and when they were recorded, and discrepancies raise immediate questions about the integrity of the entire batch record.
Health Canada’s Good Production Practices require licence holders to retain documentation demonstrating compliance with Cannabis Regulations Part 5. This includes batch records, storage records, deviation records, and calibration logs. While the regulations do not use the word “contemporaneous” explicitly, the expectation that records accurately reflect facility operations inherently requires timely recording. A batch record showing that 15 weighing activities were all entered into the system within a two-minute window at 4:47 PM, when the activities themselves were performed between 9:00 AM and 3:00 PM, tells an inspector exactly one thing: those records were entered after the fact.
Health Canada conducted 889 inspections of cannabis licence holders in FY 2024-2025, issuing 37 non-compliant reports. “Unsatisfactory retention of documents and information” was a named finding category. While the public reports do not break down findings by individual ALCOA++ principle, the pattern is clear: record-keeping deficiencies are a primary inspection target, and contemporaneous gaps are among the easiest for inspectors to detect through timestamp analysis.
The regulatory pressure extends beyond Canada. The FDA’s 21 CFR Part 11 requires computer-generated, time-stamped audit trails that record the date and time of operator entries. The revised EU GMP Annex 11 draft, expected to be finalised in mid-2026, explicitly references ALCOA+ for the first time in EU GMP text and expanded from 5 pages to 19 pages (PharmOut, 2025). For cannabis facilities pursuing EU GMP certification or international export, contemporaneous documentation is not optional.
How Do Cannabis Facilities Typically Fail the Contemporaneous Principle?
The most common failure has a name in the industry: the “notebook then re-key” workflow. It is widespread, well-understood by inspectors, and structurally impossible to fix without changing how data enters the system. This workflow also violates the Original data recording principle, since the system entry is a copy of the notebook rather than the first capture of the measurement.
The Notebook-to-Desktop Workflow
Here is what it looks like in practice. An operator on the production floor weighs a batch on a scale. The scale displays a reading. The operator writes the weight, the lot number, and the time in a paper notebook (or on a loose sheet, or on a clipboard). The operator continues working through multiple batches. At some point, often at the end of the shift or during a break, the operator walks to a desktop computer and re-enters the data into the seed-to-sale system. The system timestamp records the moment of data entry, not the moment of the weighing activity.
This workflow violates the Contemporaneous principle in at least three ways:
- Timestamp inaccuracy: The system records show the data was entered at 4:30 PM; the actual weighing happened at 10:15 AM. An inspector reviewing the audit trail sees a six-hour gap.
- Memory degradation: By the time the operator re-enters data, details may be misremembered. Was that 142.3 grams or 143.2 grams? The notebook entry might be illegible, compounding the error.
- Batch context loss: When multiple batches are recorded in a single desktop session, the sequence, timing, and context of the original activities are compressed into a data-entry session that bears no resemblance to the actual production flow.
Desktop-Only Software Does Not Solve the Problem
A common misconception is that switching from paper to a seed-to-sale software system automatically addresses the Contemporaneous principle. It does not, unless the software is accessible at the point of activity.
If an operator must leave the production floor, remove gloves, walk to a workstation in a separate room, log in, navigate to the correct screen, and enter data, the delay still exists. The timestamp is still wrong. The contemporaneous gap persists regardless of how sophisticated the software is. The location of data entry matters as much as the medium.
| Workflow | Timestamp Reflects | Contemporaneous | Risk Level |
|---|---|---|---|
| Paper notebook, re-keyed at desktop | Time of desktop entry (hours later) | Fail | High: inspector sees timestamp clusters |
| Desktop-only software, entered after activity | Time of desktop entry (minutes to hours later) | Fail | Medium: gap reduced but still detectable |
| Mobile device on production floor | Time of submission on the floor | Supported | Low: timestamp aligns with activity |
GrowerIQ Mobile Scanner
See how it supports contemporaneous data capture on the facility floor
Capture data at the moment the action happens, with timestamps generated at submit time.
Explore the Scanner →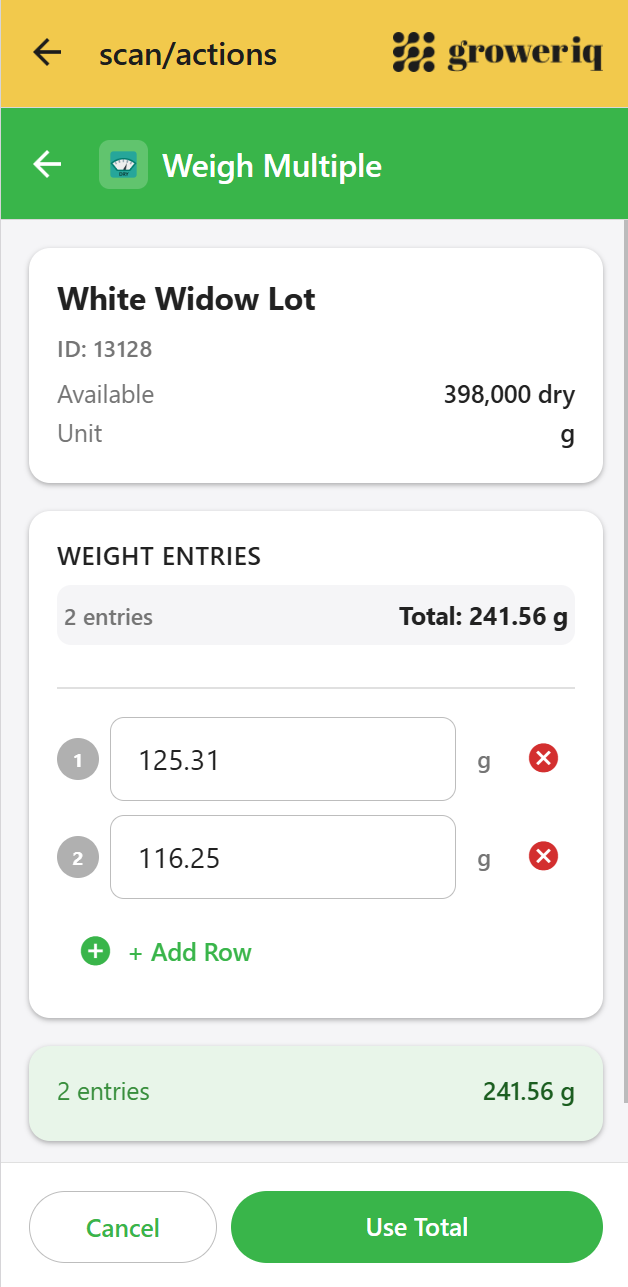
What Does a Contemporaneous Documentation Solution Look Like?
The answer is bringing the data capture device to the point of activity, so the system timestamp and the activity timestamp are the same moment.
Mobile data capture on the production floor eliminates the notebook-to-desktop workflow entirely. When an operator weighs a batch, inspects a lot, or transfers inventory, they submit the record directly from a handheld device while standing at the point of activity. The timestamp is generated at the moment of submission, not pre-filled from a prior step or entered retroactively.
How Mobile Capture Supports the Contemporaneous Principle
The mechanics matter. A well-designed mobile system generates the ISO 8601 timestamp at the moment the operator taps “Submit,” not when they open the form or scan a barcode. This means the recorded time reflects when the data was committed, closing the gap between activity and record. The submission triggers an immediate API call to the backend. There is no batch upload at end of shift, no intermediate local storage that could be modified, and no opportunity for the record to diverge from reality.
This is the area where GrowerIQ’s mobile scanner provides the most direct support for ALCOA++ data integrity objectives. The scanner is a purpose-built application designed for ruggedised Android devices (including Zebra MC3300 handhelds) that operators carry on the production floor. Data flows from the operator’s action through the form submission to the backend database in a single, uninterrupted chain. No notebooks. No re-keying. No desktop detours.
Consider the difference in a typical weighing workflow:
| Step | Notebook + Desktop Workflow | Mobile Floor Capture |
|---|---|---|
| 1 | Operator reads scale display | Operator scans lot barcode on device |
| 2 | Writes weight on paper | Weight auto-populated from connected scale (or entered on device) |
| 3 | Continues to next batch | Taps Submit; timestamp generated; API POST sent immediately |
| 4 | After multiple batches, walks to desktop | (No step 4. Record is already in the system.) |
| 5 | Re-enters all data from notebook | |
| 6 | System timestamp: 4:30 PM (entry time) | System timestamp: 10:15 AM (activity time) |
The three-step mobile workflow produces a record that an inspector can trust: the timestamp matches the activity, the data entered the system without intermediate transcription, and the submission is individually attributable to the logged-in operator.
Beyond Weighing: Every Floor Activity Benefits
The Contemporaneous principle applies to every recorded activity, not just weighing. Visual inspections, inventory transfers, packaging counts, environmental checks, and deviation flags all need to be recorded when they happen. A mobile device on the production floor makes contemporaneous recording the default workflow rather than an aspiration that depends on operator discipline.
Validation Disclaimer: ALCOA++ data integrity is achieved through validated processes at your facility. GrowerIQ provides the software foundation that supports the Contemporaneous principle; your quality team completes the validation documentation (IQ/OQ/PQ) as part of your facility’s quality management system. For guidance on the validation process, see our post on GMP software validation for cannabis.
Key Takeaways
- Contemporaneous means “at the time of activity”: PIC/S PI 041-1 and WHO TRS 996 both require data to be recorded when the action happens, not after the fact.
- The “notebook then re-key” workflow is the most common violation: Any delay between performing an activity and entering it into the system creates a timestamp discrepancy that inspectors can detect.
- Desktop-only software does not fix the gap: If operators must leave the production floor to enter data, the Contemporaneous principle is still violated regardless of the software’s capabilities.
- Mobile capture closes the gap at its source: Bringing data entry to the point of activity aligns the system timestamp with the actual activity time.
- Software is the foundation, not the finish line: Facility-level validation (IQ/OQ/PQ) by your quality team completes the ALCOA++ data integrity picture.
Frequently Asked Questions
What does contemporaneous documentation mean in cannabis GMP?
Contemporaneous documentation means recording data at the time the activity is performed. In a cannabis facility, this applies to every recorded action: weighing, inspecting, transferring, and packaging. The system timestamp should reflect when the activity happened, not when an operator later entered it at a desktop. PIC/S PI 041-1 states that “backdating or post-hoc recording undermines the integrity of the record.”
How do inspectors detect contemporaneous documentation failures?
Inspectors examine the audit trail for timestamp patterns. If a batch record shows 20 activities all entered within a five-minute window at the end of a shift, but the activities themselves span an eight-hour production day, the timestamps reveal after-the-fact data entry. This pattern is easy to identify in any system with audit trail capability, and it raises immediate questions about the reliability of the entire record.
Does Health Canada require contemporaneous documentation for cannabis?
Health Canada’s Good Production Practices do not use the word “contemporaneous” explicitly, but the Cannabis Regulations (Part 5) require documentation that demonstrates compliance with production practices. The expectation that records accurately reflect facility operations inherently requires timely recording. ALCOA++ (including the Contemporaneous principle) represents the pharmaceutical best-practice framework for meeting those requirements.
Can desktop-only seed-to-sale software meet the contemporaneous requirement?
Not fully. If operators must leave the production floor to enter data at a workstation, there is always a delay between the activity and the record. The system timestamp reflects entry time, not activity time. Mobile data capture on the production floor is the most effective way to close this gap, because the device is present at the point of activity.
Last updated: March 2026
Capture Data at the Moment It Happens
GrowerIQ’s mobile scanner brings data capture to the production floor, supporting the Contemporaneous principle and all 10 ALCOA++ data integrity objectives. The first seed-to-sale mobile scanner with native live scale integration.
EXPLORE SCANNER
About GrowerIQ
GrowerIQ is changing the way producers use software - transforming a regulatory requirement into a robust platform to learn, analyze, and improve performance.
To find out more about GrowerIQ and how we can help, fill out the form to the right, start a chat, or contact us.

