When an inspector asks for the batch record from lot #4471, how many minutes does it take to find it?
Accessible records cannabis GMP audit readiness depends on a deceptively simple requirement: every batch record, deviation report, and calibration log must be retrievable without delay throughout the full retention period. The Available principle is the ninth element of the ALCOA++ data integrity framework, and it addresses a problem that paper-based facilities discover only when an inspector is standing in the room. Records that exist but cannot be found are, from a regulatory standpoint, records that do not exist.
This guide defines what “Available” means under international GMP standards, explains why instant retrieval matters during Health Canada inspections, identifies where cannabis facilities most commonly fall short, and outlines how digital record management closes the gap. This article is part of our comprehensive ALCOA++ Data Integrity for Cannabis Manufacturing guide.
What Does “Available” Mean for Cannabis Data Integrity?
PIC/S PI 041-1, the definitive international guidance on data integrity published by the Pharmaceutical Inspection Co-operation Scheme (whose member authorities include Health Canada), defines the Available principle as follows: “Information should be readily accessible and available for review or inspection throughout the retention period.” The emphasis is on “readily.” A record that takes 45 minutes to locate, or one that requires a specific employee’s knowledge of where it was filed, does not meet the standard.
The Available principle differs from the Enduring principle in an important way. Enduring asks whether a record will survive intact for the full retention period. Available asks whether that surviving record can be found and produced when someone needs it. A paper batch record locked in a fireproof cabinet is enduring; the same record misfiled in the wrong binder is not available. Both principles must be satisfied simultaneously, and a failure on either one compromises the record’s regulatory value.
Retrieval also depends on traceability. If a batch record cannot be traced through its lifecycle, from harvest through processing through packaging, finding the complete record set for a given lot requires manual reconstruction rather than a single query. The Available, Enduring, and Traceable principles work as an interconnected triad: records must persist, be locatable, and be linked.
Why Does Record Availability Matter During Health Canada Inspections?
Because the speed of retrieval signals the quality of your entire record-keeping system, and inspectors know it.
Health Canada’s Cannabis Regulations (Part 5, Sections 231-232) require licence holders to retain documentation demonstrating compliance with Good Production Practices and Quality Assurance requirements. This includes batch records, storage records, temperature and humidity logs, deviation records, calibration records, and complaint investigation files. Each version must be retained for at least two years after it is replaced or after the licence expires. Health Canada’s GPP Guide for Cannabis sets detailed expectations for what must be available on request.
During an on-site inspection, an inspector may request records for a specific lot at any point in the visit. The request is rarely anticipated. An inspector reviewing your drying room might ask for the harvest record of a batch currently on the racks. A walkthrough of your packaging area might prompt a request for the processing and QA inspection records of the lot being packaged. The ability to produce those records promptly, within seconds rather than tens of minutes, demonstrates that your record-keeping system is functioning as intended. Conversely, delays in retrieval raise questions about whether the records exist at all, whether they might have been altered, or whether the facility’s documentation practices are reliable enough to support the licence.
The enforcement context reinforces this point. Health Canada’s compliance and enforcement data confirms that documentation deficiencies remain a persistent finding category in cannabis inspections (see our complete ALCOA++ guide for the latest enforcement figures). Record availability failures, where records technically exist but cannot be produced during an inspection, fall squarely within that category.
Where Do Cannabis Facilities Fail at Accessible Records During GMP Audits?
Record availability failures cluster into four patterns, each rooted in the physical limitations of paper or the organisational limitations of poorly structured digital systems.
1. The Filing Cabinet Problem
Paper batch records are stored in binders, organised by date, lot number, or production area. Over months and years, binders accumulate. Records get misfiled. Pages are removed for photocopying and never returned. When an inspector requests a specific batch record, an employee must physically search through dozens of binders to locate it. If the employee who originally filed the record is not on shift, the search takes even longer. In some cases, the record is never found.
2. Single-Point-of-Knowledge Retrieval
Many cannabis facilities depend on one or two people who “know where everything is.” The quality manager keeps a mental map of which binder holds which records, or the production lead maintains a personal spreadsheet that cross-references lot numbers to filing locations. When that person is absent, on vacation, or has left the company, the facility’s ability to retrieve records degrades immediately. Accessible records cannabis GMP audit readiness cannot depend on any single individual’s institutional knowledge.
3. Fragmented Digital Records
Facilities that have partially digitised their records often end up with a worse retrieval problem than pure paper operations. Some records live in the seed-to-sale platform, others in spreadsheets on a shared drive, others in scanned PDFs stored on a local machine, and still others on paper in a binder. An inspector requesting the complete record set for a lot may need to search four different systems and one filing cabinet. The time to reconstruct the complete picture can be substantial.
4. Destroyed or Degraded Records
Records that no longer exist are, by definition, not available. Paper records stored in production environments are vulnerable to water damage, chemical exposure, and physical deterioration. Thermal paper from scale printers fades within months. USB drives containing exported spreadsheets can fail without warning. Records stored on decommissioned computers may be lost entirely. This failure overlaps with the Enduring principle, but the regulatory consequence is felt at the Available level: the inspector asked for a record, and it could not be produced.
| Failure Pattern | Root Cause | Inspector Impact |
|---|---|---|
| Filing cabinet delays | Paper records organised manually; misfiling common | Inspector waits 15-45 minutes; questions overall record quality |
| Single-point-of-knowledge | Retrieval depends on one person’s institutional memory | Records effectively unavailable when that person is absent |
| Fragmented digital records | Data split across multiple systems with no unified search | Complete record set requires assembling pieces from four or five sources |
| Destroyed or degraded records | Paper, thermal prints, or unbackuped digital media fail over time | Record cannot be produced at all; regulatory gap documented |
GrowerIQ Mobile Scanner
See how it supports instant record retrieval on the facility floor
Digital records stored in PostgreSQL, retrievable in seconds during any inspection.
Explore the Scanner →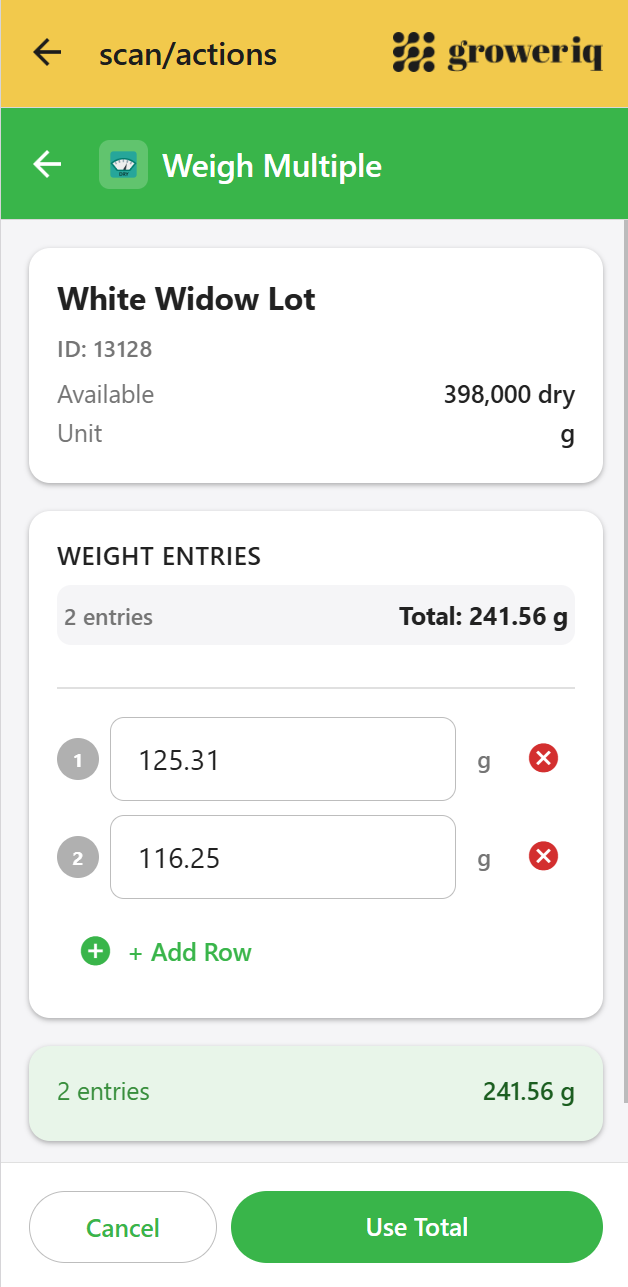
How Do You Support Accessible Records for Cannabis GMP Audits?
The solution is a single, searchable, always-on system of record. Every batch record, every activity, and every audit trail entry stored in one place, retrievable by any authorised user within seconds.
Centralised Database Storage
When all operational records are stored in a relational database (such as PostgreSQL), retrieval becomes a query rather than a search. An inspector asks for the batch record for lot #4471. A staff member types the lot number into a search field, and the complete record set appears: harvest activity, drying log, processing steps, QA inspections, packaging records, and the full audit trail of every modification. No binders, no cross-referencing, no dependence on a specific employee’s filing knowledge.
GrowerIQ stores all activity records in PostgreSQL, retrievable through the web application’s API at any time. Every batch record, inventory movement, quality inspection, and audit trail entry is indexed and searchable by lot number, date range, operator, activity type, or any combination of these fields. The complete history of a batch is accessible from any device with a browser and appropriate permissions.
Instant Retrieval During Inspections
The practical test of record availability is the inspection scenario. An inspector points at a batch on your production floor and asks for its complete history. With digital records in a centralised database, a staff member can pull up the full record set in seconds. With paper records, the same request initiates a physical search that may take tens of minutes and may not produce the complete record.
Digital retrieval also eliminates the knowledge-dependency problem. Any authorised user, whether it is the quality manager, a production supervisor, or a compliance officer, can access the same records through the same interface. The system does not depend on one person knowing which binder to check.
Access From Any Device
A web-based system means records are accessible from any device with a browser and network connectivity: a desktop in the QA office, a tablet on the production floor, or a laptop in a conference room during an inspection meeting. This flexibility removes the physical constraint of paper records, which require someone to be in the room where the binders are stored.
For floor operations, GrowerIQ’s mobile scanner provides read access with brief connectivity resilience. The scanner caches recently viewed data (with a five-minute freshness window and a ten-minute retention period), so operators can review inventory and batch details even during momentary network interruptions. Write operations (recording new activities) require active network connectivity and provide immediate feedback if the connection is unavailable, with a retry option built into the interface.
No Records Lost, Misfiled, or Destroyed
Digital records in a managed database are not subject to the physical vulnerabilities that plague paper. They cannot be water-damaged, misfiled, accidentally discarded, or lost when an employee leaves the company. Combined with the Enduring principle’s requirements for permanent storage and immutable audit trails, centralised digital storage ensures that records remain both intact and findable for the full retention period.
Paper vs Digital: How Does Each Approach Handle Record Availability?
The gap between paper and digital record availability is not a matter of degree. It is a structural difference in how retrieval works.
| Availability Requirement | Paper-Based | Digital (Centralised Database) |
|---|---|---|
| Retrieval speed | Minutes to hours. Physical search through binders; depends on filing accuracy. | Seconds. Database query by lot number, date, operator, or activity type. |
| Knowledge dependency | High. Often requires specific employee who knows the filing system. | None. Any authorised user queries the same searchable interface. |
| Complete record assembly | Manual. Staff must locate and collate pages from multiple binders or locations. | Automatic. All records for a batch returned in a single query. |
| Remote access | Not possible. Physical records require presence in the storage location. | Supported. Any device with a browser and network access. |
| Resilience to staff turnover | Low. Institutional knowledge of filing system leaves with the employee. | High. Records and retrieval interface persist regardless of personnel changes. |
| Risk of permanent loss | High. Paper degrades, gets misfiled, or is accidentally discarded. | Low. Database with backups; records indexed and searchable indefinitely. |
The critical insight is that paper availability depends entirely on human systems: consistent filing, accurate cross-referencing, and institutional knowledge retention. Digital availability depends on system architecture: a searchable database, structured queries, and role-based access controls. Human systems degrade over time as staff turn over and filing volumes grow. System architecture scales without degradation.
Key Takeaways
- Available means retrievable without delay: PIC/S PI 041-1 requires that records be readily accessible throughout the retention period, not just preserved but findable on demand.
- Retrieval speed signals record quality: When an inspector requests a batch record, delays of minutes or tens of minutes raise questions about the entire documentation system.
- Four failure patterns dominate: Filing cabinet delays, single-point-of-knowledge dependency, fragmented digital records, and physical record destruction are the most common barriers to accessible records cannabis GMP audit readiness.
- Centralised digital storage solves the structural problem: A searchable database eliminates physical search, removes knowledge dependency, and provides instant retrieval from any authorised device.
- Software is the foundation, not the finish line: Facility-level validation (IQ/OQ/PQ) by your quality team completes the record availability picture.
Frequently Asked Questions
What does accessible records cannabis GMP audit readiness actually require?
It requires that any record an inspector requests, whether a batch record, deviation report, calibration log, or audit trail, can be produced without significant delay. PIC/S PI 041-1 uses the term “readily accessible,” meaning retrieval should be measured in seconds, not minutes. The system should not depend on a specific employee’s knowledge of where records are stored.
How long must cannabis records be retained and available under Health Canada regulations?
Cannabis Regulations Part 5 (Sections 231-232) requires licence holders to retain each version of documentation for at least two years after the version is replaced or the licence expires. During that entire retention period, the records must be available for inspection. This applies to batch records, storage logs, deviation records, calibration records, and complaint investigation files.
Can digital records satisfy the Available principle if the system goes offline?
Temporary system outages are a practical reality. The key is that records remain accessible throughout the retention period under normal operating conditions, and that the system has appropriate backup and disaster recovery measures. For on-floor operations, read caching provides brief resilience to connectivity interruptions, allowing operators to view recent data during momentary network gaps. Write operations require active connectivity and provide immediate user feedback if the connection is unavailable.
How does the Available principle relate to the Enduring and Traceable principles?
The three principles form an interconnected triad. Enduring ensures the record survives intact for the full retention period. Available ensures the surviving record can be found and produced when needed. Traceable ensures that related records are linked so that the complete record set for a batch can be retrieved as a unit rather than assembled manually. A failure in any one of these three principles undermines the other two.
Validation Disclaimer: ALCOA++ data integrity is achieved through validated processes at your facility. GrowerIQ provides the software foundation that supports each ALCOA++ principle; your quality team completes the validation documentation (IQ/OQ/PQ) as part of your facility’s quality management system.
Last updated: March 2026
See How Digital Record Management Supports Instant Retrieval During GMP Audits
GrowerIQ stores every batch record, activity, and audit trail entry in a searchable PostgreSQL database, retrievable by any authorised user from any device.
EXPLORE SCANNER
Recommended For You
HighIQ Webinar Series: Powering Precision Production with Total Grow Control
May 8, 2026Portugal’s Cannabis Exports Triple: 42 Tonnes to Germany and Growing as Europe’s Processing Hub
April 22, 2026UK Medical Cannabis Prescriptions Surge 262%: 80,000 Patients and a GBP 500M Private Market
April 21, 2026About GrowerIQ
GrowerIQ is changing the way producers use software - transforming a regulatory requirement into a robust platform to learn, analyze, and improve performance.
To find out more about GrowerIQ and how we can help, fill out the form to the right, start a chat, or contact us.

